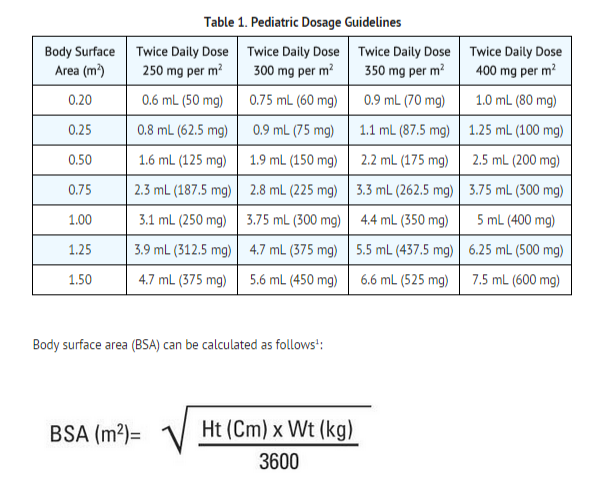
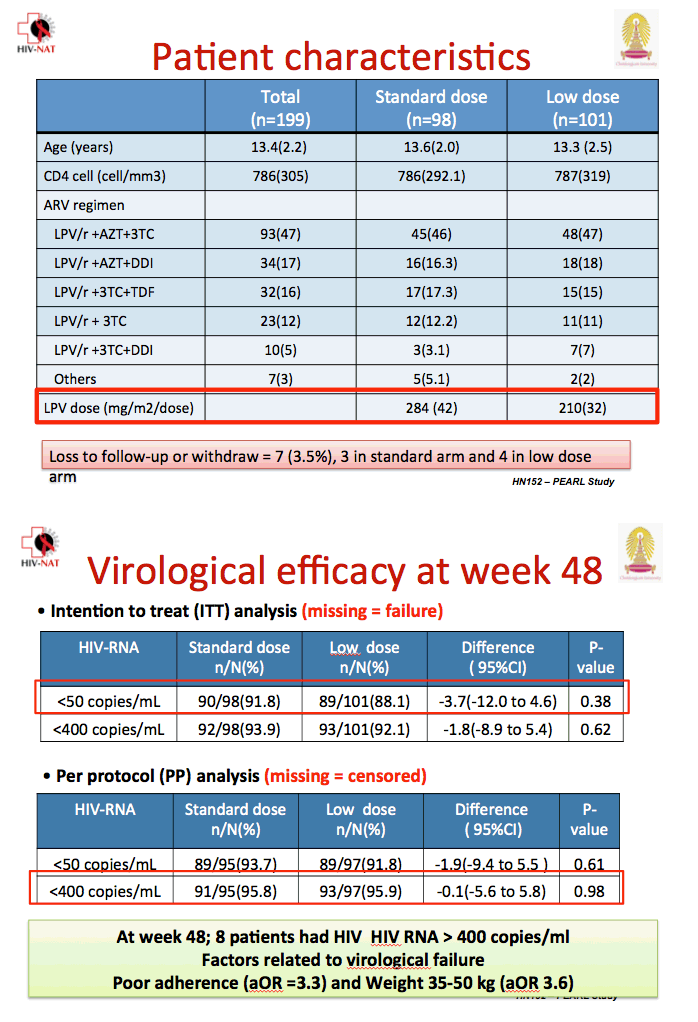
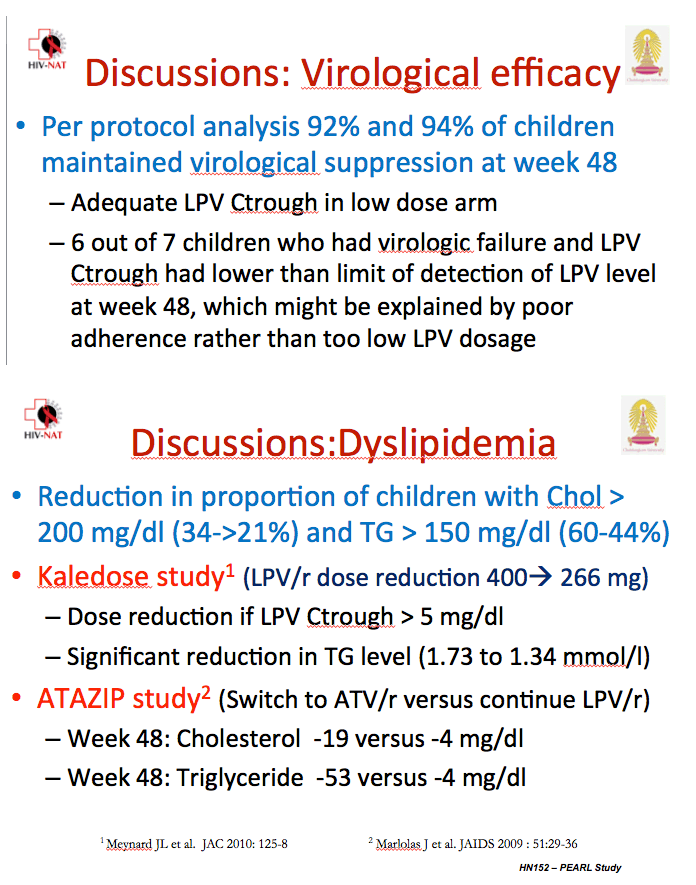
![PDF] Lopinavir/Ritonavir plus Lamivudine and Abacavir or Zidovudine dose Ratios for Paediatric Fixed-Dose Combinations | Semantic Scholar PDF] Lopinavir/Ritonavir plus Lamivudine and Abacavir or Zidovudine dose Ratios for Paediatric Fixed-Dose Combinations | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/2c44d97f8a986b4311c9e3cdf61ff734036a4cd7/14-Table1-1.png)
PDF] Lopinavir/Ritonavir plus Lamivudine and Abacavir or Zidovudine dose Ratios for Paediatric Fixed-Dose Combinations | Semantic Scholar

Reduced dose darunavir/ritonavir effective and safe for people switching while virally suppressed | aidsmap
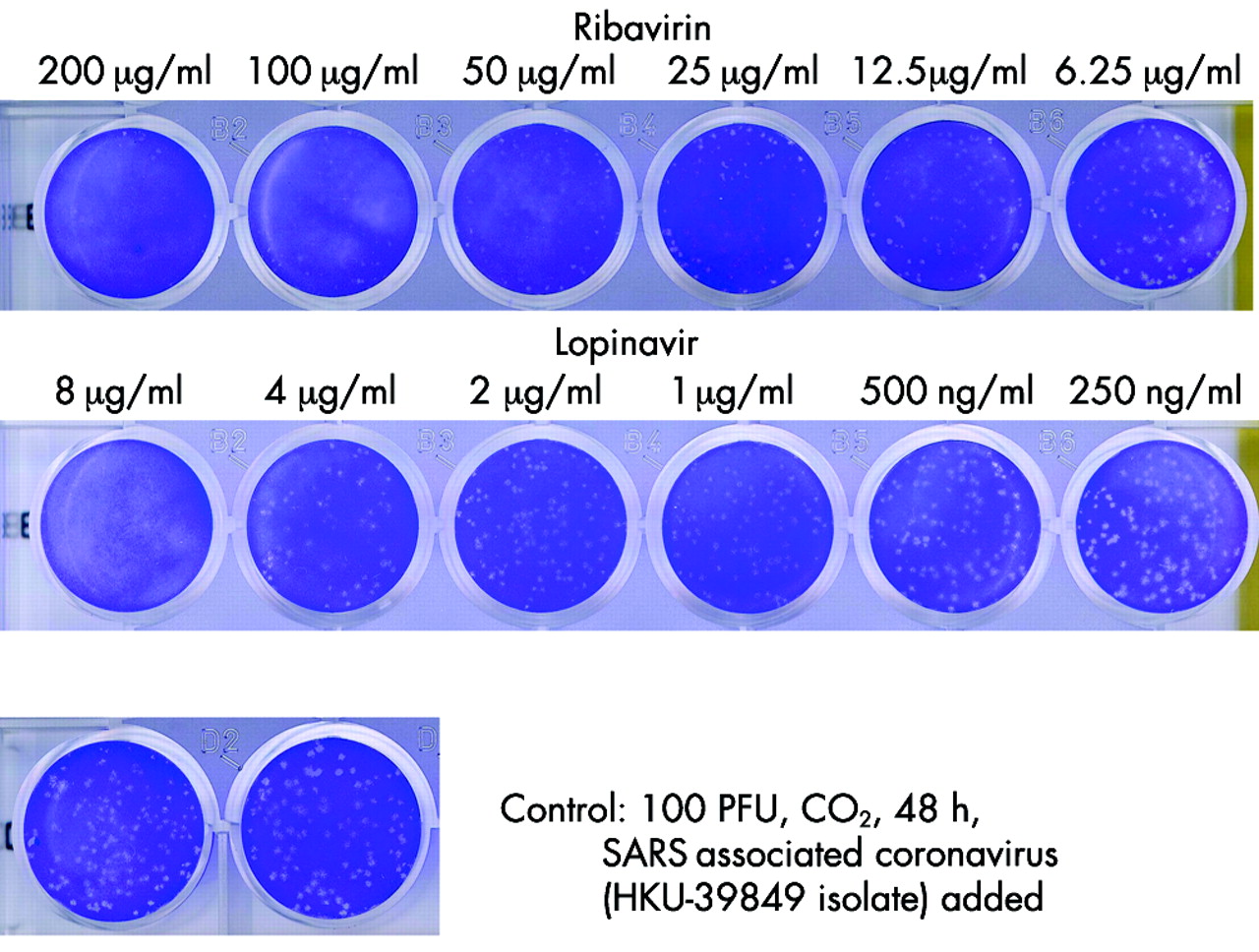
Role of lopinavir/ritonavir in the treatment of SARS: initial virological and clinical findings | Thorax

Ritonavir/Lopinavir and Its Potential Interactions With Psychiatric Medications: A COVID-19 Perspective | Psychiatrist.com
Randomised Pharmacokinetic Trial of Rifabutin with Lopinavir/Ritonavir-Antiretroviral Therapy in Patients with HIV-Associated Tuberculosis in Vietnam | PLOS ONE

Long-Term Treatment with Lopinavir-Ritonavir Induces a Reduction in Peripheral Adipose Depots in Mice | Antimicrobial Agents and Chemotherapy
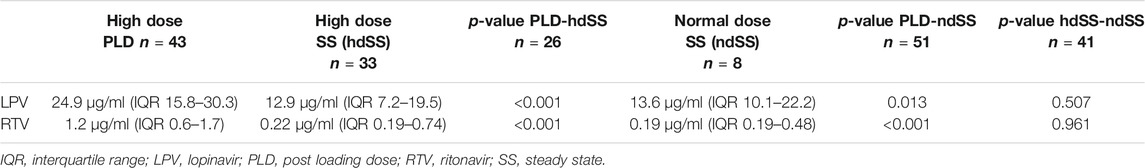
Frontiers | High Dose Lopinavir/Ritonavir Does Not Lead to Sufficient Plasma Levels to Inhibit SARS-CoV-2 in Hospitalized Patients With COVID-19
LOPINAVIR/RITONAVIR MYLAN 1. Product Name 2. Qualitative and Quantitative Composition 3. Pharmaceutical Form 4. Clinical Particu
Randomised Pharmacokinetic Trial of Rifabutin with Lopinavir/Ritonavir-Antiretroviral Therapy in Patients with HIV-Associated Tuberculosis in Vietnam | PLOS ONE

Low-dose ritonavir-boosted darunavir once daily versus ritonavir-boosted lopinavir for participants with less than 50 HIV RNA copies per mL (WRHI 052): a randomised, open-label, phase 3, non-inferiority trial - The Lancet HIV

Lopinavir metered-dose transdermal spray through microporated skin: Permeation enhancement to achieve therapeutic needs - ScienceDirect